Abstract
Recognition of hereditary hematopoietic malignancies (HHMs) has accelerated in recent years, with deleterious germline variants in more than 20 genes recognized as drivers of myeloid malignancies. Individuals with HHMs generally receive similar treatment plans as those with de novo disease. Hematopoietic stem cell transplantation (HSCT) is often employed as a curative approach for myeloid malignancies, and in those with germline pathogenic (P)/likely pathogenic (LP) predisposition variants, there is a preference for use of related donors who lack the familial variant. However, there are limited data on the outcomes of patients with HHMs who are treated in this manner. In this study, we used comprehensive clinical and genomic data to investigate the allogeneic transplant-related outcomes in the largest international myeloid HHM cohort reported to date.
We performed a retrospective analysis of all patients with myeloid malignancies who underwent germline predisposition testing at The University of Chicago (UofC) or the Central Adelaide Health Network between 2010 and 2021; or were known to have deleterious germline DDX41 variants from the University of Pennsylvania and Moffitt Cancer Center.
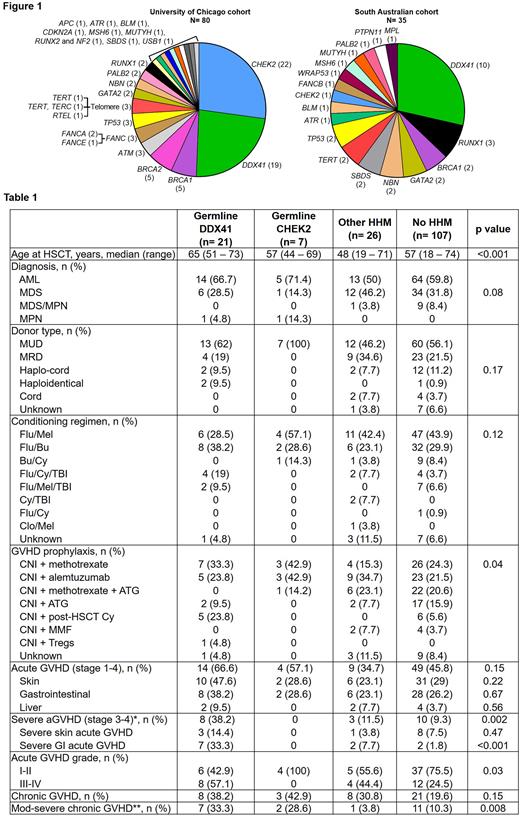
We studied 472 patients, of whom 28% (80/289) in the UofC cohort and 20% (35/175) in the Adelaide cohort had a deleterious germline variant causative of HHMs. In the UofC cohort, P/LP CHEK2 variants were the most common, whereas P/LP DDX41 variants were most common in the Adelaide cohort (Fig. 1). A total of 161 patients (34% of the entire cohort) underwent allogeneic HSCT for their myeloid neoplasm (Table 1). There were no differences in the distribution of diagnoses, donor types, disease status at the time of HSCT (remission vs active disease), conditioning regimens, or conditioning intensity. All donor cells were mobilized from peripheral blood. Almost all donors lacked the deleterious germline causative variant, with one donor sharing a deleterious familial DDX41 variant with the relative/HSCT recipient. The median time to engraftment was similar between those who had vs lacked germline HHM variants.
Patients with P/LP germline DDX41 variants had higher rates of severe (stage 3-4) acute graft versus host disease (GVHD) when compared to patients with P/LP CHEK2, those with other germline variants, or those without P/LP germline variants (38% vs 0, 12%, and 9%, respectively; p=0.002). Notably, severe gastrointestinal GVHD was seen in a third of patients with P/LP germline DDX41 variants as opposed to <10% incidence in other groups (p<0.001, Table 1). Similarly, 33% of patients with P/LP germline DDX41 variants experienced moderate to severe chronic GVHD, whereas this incidence was 29% for patients with P/LP germline CHEK2 variants, 4% for patients with other P/LP germline variants, and 10% for patients without a P/LP inherited variant (p=0.008). Patients with P/LP germline DDX41 variants who received post-HSCT cyclophosphamide had lower rates of severe acute GVHD compared to those who received other forms of GVHD prophylaxis (0% vs 53%, p=0.03). To adjust for the effects of known GVHD risk factors, we performed a multivariable logistic regression analysis. Germline DDX41 status remained an independent predictor of both acute GVHD (OR: 4.76, range 1.27-17.9; p=0.01) and chronic GVHD (OR: 3.39, range 1.09-10.5; p=0.03). Importantly, post-HSCT cyclophosphamide use was associated with a reduced risk of acute GHVD when adjusted for other variables (OR: 0.10, range 0.01-0.71; p=0.008).
Our data establish severe GVHD as a clinically relevant post-transplant phenomenon in germline DDX41-associated HHMs and suggest that donor-derived wild-type T-cells may become activated by a pro-inflammatory milieu conferred by P/LP germline DDX41 variants. Moreover, our observations suggest that post-HSCT cyclophosphamide is an effective strategy for GVHD prevention in these individuals. Broader confirmation of our results in other heterogeneous patient cohorts will serve to expand the spectrum of known pathogenic variants implicated in HHMs and support the development of targeted interventions for those with deleterious germline HHM alleles. Finally, these results argue for germline genetic testing in patients prior to allogeneic HSCT.
Disclosures
Gill:Astra Zeneca: Honoraria; Immpact Bio: Honoraria; Mission Bio: Membership on an entity's Board of Directors or advisory committees; Hemogenyx: Honoraria, Research Funding; Asher Bio: Research Funding; Carisma: Current holder of stock options in a privately-held company, Research Funding; Interius: Current holder of stock options in a privately-held company, Research Funding; Novartis: Patents & Royalties, Research Funding. Elmariah:Bristol Myers Squibb: Research Funding. Bishop:WindMIL Therapeutics: Consultancy; Celgene: Honoraria; Incyte: Honoraria, Other: Travel support , Speakers Bureau; Tmunity: Research Funding; Iovance: Consultancy; Bluebird Bio: Consultancy; CRISPR Therapeutics: Consultancy, Research Funding; Sanofi: Honoraria, Speakers Bureau; Kite, a Gilead Company: Consultancy, Honoraria, Other: Travel support, Research Funding, Speakers Bureau; Bristol Myers Squibb: Honoraria, Other: Travel support, Speakers Bureau; Autolus: Consultancy, Research Funding; Chimeric Therapeutics: Consultancy; Sana Biotechnology: Consultancy; ADC Therapeutics: Speakers Bureau; Triumvira: Research Funding; Immatics: Research Funding; Agios: Consultancy, Honoraria, Other: Travel support, Speakers Bureau; Servier: Speakers Bureau; Arcellx: Consultancy, Research Funding; Novartis: Consultancy, Honoraria, Other: Travel support , Research Funding. Hiwase:AbbVie: Speakers Bureau; Novartis: Speakers Bureau; BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.